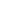What Are Transition Metals? Definition, Properties, and Uses
 Published:Feb 27,2026
Published:Feb 27,2026

Transition metals are from the various chemical elements that possess valance electrons. These elements are present in the middle portions of periodic table. The term “transition” is not related to any specific chemical significance. But it is used to distinguish the similarities of atomic structures and properties of those elements. This article will give you deep insights into the behavior, properties and applications of these transition elements.
What Are Transition Metals?
Transition metallic elements have atoms that have a partially filled d-subshell. These can form stable cations with an incompletely filled d-subshell. The transition metal series shows variable oxidation states. They form colored compounds and serve as catalysts in chemical reactions.
Where Are the transition Metals on the Periodic Table?
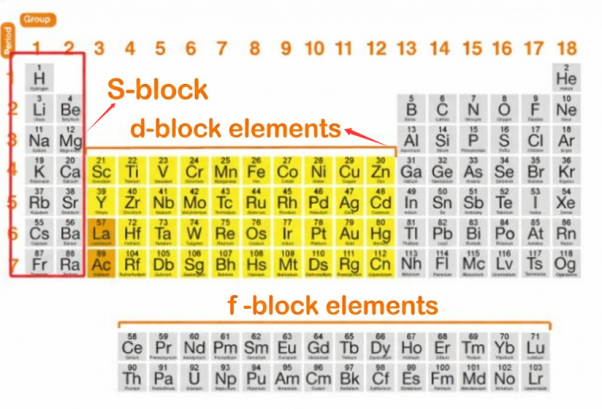
Transition metals are found in the d-block of the periodic table. They cover central section between Group 3 and 12 and period 4,5,6 and 7. D-block has four main transition metal series. These are 3d (Sc-Zn), 4d (Y-Cd), 5d (La-Hg), and 6d (Ac-Cn). The inner transition metals contain 14 elements after Lanthanum, and 14 elements after Actinium. They are referred as “f-block elements “
Are Transition Metals Always Magnetic?
No, transition metals are not always magnetic in their elemental form. Most transition metals are paramagnetic. This means they are weakly attracted to a magnetic field. This is caused by the presence of unpaired electrons in their (n-1) d-orbital. But certain transition metals have filled or completely empty d-orbitals in specific compounds. These are diamagnetic like Cu+ or Sc3+.
Are Transition Metals Reactive?
Yes, transition metals are reactive. They have relatively reaction metals like Ti or Fe to extremely unreactive "noble" metals like Gold or silver. The reactivity is defined by the electron configuration and their atomic stability. Reactivity influences material selection and processing of the elements of transition metals series.
For example, some metals like Chromium or Nickle have their own protective layer and provide better corrosion resistance, while non-reactive metal like gold or silver are used as protective coating on more reactive compounds. Similarly, high reactive metals need specialized coolants during CNC machining to prevent oxygen reaction at high temperatures.
Why Are Transition Metals Called “Transition” Metals?
Transition metals show “transition" in electron configuration and chemical properties. like they have highly reactive metals on the left side of the periodic table (s-block) and the non-metals on the right side (p-block).
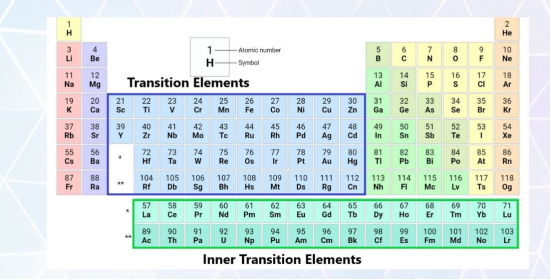
Are Transition Metals Metalloids?
No, transition metals are not metalloids. Transitions metals are located in the center d-block. They are shiny, ductile and are excellent conductors of electricity and heat. while metalloids are located along a zigzag line separating metals and non-metals. They have properties intermediate between metals and non-metals and often are semiconductors.
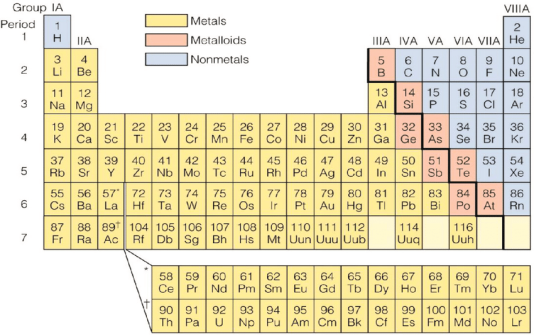
Transition Metals vs Metalloid
The difference between the elements of transition metals series and metalloids are as:
|
Feature |
Transition Metals (d-block) |
Metalloids (Semimetals) |
|---|---|---|
|
Location |
Center portion, Groups 3-12 |
Diagonal and Groups 13-16 |
|
Properties |
High density, hard, shiny |
Brittle, shiny and dull |
|
Conductivity |
Excellent conductor |
Fair conductor |
|
Color |
colored compounds |
colorless |
|
Examples |
Iron, Copper, Gold, Silver, Nickel |
Silicon, Germanium, Arsenic, Boron |
What Are Properties of Transition Metals?
Transition metal elements are characterized by their physical properties. For example, high melting points, densities, and hardness and strength compared to alkali metals. These are highly due to strong metallic bonding. Some notable metals like Cu, Ag, and Au are excellent conductors of heat and electricity.
High Melting Points
Most have high melting points and boiling points due to strong metallic bonds. The bond between s and d electrons increases the melting temperature by more than 1000◦C. But there is a notable exception of mercury, which is liquid at room temperature. Typically, Tungsten has the highest melting point.
High Density
Transition metal elements are much denser than other metals. These are metals normally in groups 1 and 2, because their atoms are closely packed and have a high mass-to-volume ratio.
Hard and Strong
Transition metals show high great hardness and tensile strength. Therefore, they are more durable for structural uses and alloys like steel.
Good Thermal Conductivity
Transition metals series have superior conduction of heat because of their large number of delocalized electrons. Silver and Copper are the most conductive elements among them.
Good Conductivity
Transition metals are excellent electrical conductors. Their electrical conductivity varies. Like Copper, Silver, and Gold are superior to metals like Iron and Titanium.
What Are the Common Transition Metals?
Common d-block transition metals include iron (Fe), copper (Cu), titanium (Ti), and manganese (Mn). These are found in the d-block Groups 3-12, of the periodic table.
Titanium (Ti)
Titanium is known for its high strength-to-weight ratio and corrosion resistance. It is one of the important materials used in aerospace and medical implants due to its biocompatibility. Additionally, titanium car parts are common.
Manganese (Mn)
Manganese is essential for steel production. It improves hardness and strength of the structure. Mn also serves as a crucial biological nutrient.
Copper (Cu)
Coppes is not magnetic and highly valued for its superior electrical conductivity. It is the primary material used for electrical wiring and plumbing.
Iron (Fe)
Iron in the most abundant transition metal on Earth by weight. It is the foundation of the construction industry. Iron is also the key component of hemoglobin for oxygen transport in blood.
How to Choose Transition Metals for Applications?
There are many factors to choose the right transition metal for applications. It depends on matching specific material properties to the requirements while balancing cost and manufacturing limitations.
Identify Required Properties
To match the properties with the applications, need to know the behaviors of transition metals. Like for high structural strength and hardness, iron, steel and titanium are suitable. Tungsten and molybdenum are suitable for high temperature applications. Copper has high electrical conductivity. And titanium and Nickle are best for corrosion resistance.
Manufacturability of Metals
Manufacturability is related to production feasibility. Gold and silver are highly malleable, and titanium is highly formable and ductile. They are easier to shape into complex and thin components. Iron and nickel have good weldability. But titanium needs inert gas welding to avoid oxidation. Similarly, Titanium, tungsten, and Nickel-base superalloys are difficult to machine. And copper and carbon steel are easy to machine.
Consider Cost
Cost determines the factors like high-volume production, cost-sensitivity, or specialized applications of the material. Iron and copper are affordable choices for mass-market infrastructure. Nickel and chromium are also reasonably priced. But Platinum, palladium, and rhodium are very expensive due to rarity. Titanium is also affordable, but its difficult machining process increases its final cost.
What Are Transition Metals Used For?
Transition metals use in many industries because they are tough, durable, and can be alloyed to improve specific properties like corrosion resistance.
Catalysis
Transition metals are used to speed up reactions without being consumed. Because they have partially filled d-orbitals with multiple oxidation states. Platinum and Palladium are used in automotive catalytic converters, Iron in the Haber process and Nickel used in hydrogenation.
Aerospace Components
These are chosen for high strength-to-weight ratio and can withstand extreme temperatures. Titanium is used in jet engines, airframes, and landing gears and Niobium is used in rockets.
Automotive Parts
Transition metals are preferred for their durability, corrosion resistance, and heat resistance. Stainless steel is employed in exhaust systems and Cobalt-alloy steel is used in gears components.
Medical Devices
D-block transition metals have biocompatibility, strength, and resistance to bodily fluids. Titanium is mostly used for orthopedic implants and dental implants. Also, Platinum in cancer drugs and Silver is preferred for antimicrobial coatings.
Robotic Components
Transition metals are needed for high precision, durability, and strength. High-strength steel or titanium are used in robotic joints/limbs, Nickel-based alloys are employed for structural stability in hot environments.
Custom Prototyping
Transition metals have excellent machinability and durability in prototype testing. Machined stainless steel is used in brackets, brass components, Titanium or Aluminum-Scandium alloy parts are used in custom prototyping.

Are Transition Metals Easy to Machine?
Transition metals are not uniformly easy to machine. Their machinability varies widely based on their hardness, ductility, thermal conductivity, and chemical reactivity. Some are easy to cut like copper. While some are notoriously difficult like titanium. They need specialized techniques in CNC machining.
Transition Metals Easy to Machine
Metals that are easy to machine are Copper, Brass, and many low-alloy steels. They have properties that allow for high cutting speeds, lower tool wear, and good surface finishes. These properties are low hardness and high ductility and good thermal conductivity. Low alloy steel are free-machining grades. They produce small, brittle chips that break easily and prevent chip entanglement in CNC machines.

Transition Metals Difficult to Machine
Titanium and Inconel are difficult to machine. Because they have high strength-to-weight ratios, high hardness, or high reactivity. Titanium is also a poor heat conductor. During CNC machining, the heat generated stays at the cutting tool edge. This causes rapid thermal damage and increased tool wear. At higher temperatures, titanium is also reactive. Chips can weld onto the cutting tool and cause premature tool failure.
What Are Advantages of Using Transition Metals?
In CNC machining, transition metals like steel, titanium, and copper are valued for their versatility and durability. Their use offers three primary advantages:
Excellent Alloying Capability
Transition metals elements have unique electronic structures. This makes them react easily to other elements. This capability creates customized alloys tailored for specific machining needs like better hardness or heat dissipation.
Great Mechanical Properties
Most transition metals are naturally hard and strong. They also have high melting points. These features allow CNC-machined parts to maintain their shape and integrity under the high speed, heat, and pressure of the milling process.
Corrosion Resistance
Metals like stainless steel and titanium, naturally resist rust and chemical decay. This is very important for CNC parts used in harsh environments. The applications include aerospace, marine, or medical applications.
Transition Metals vs Other Metals
Transition metals are characterized by partially filled d-orbitals, variable oxidation states, and catalytic properties. the d-block metals differ from s-block metals and post-transition metals.
D-Block Metals vs S-Block Metals
D-block metals are hard and dense. while s-block elements are soft and less dense metals, located in Groups 1 & 2. D-blocks have higher melting points relatively than s-blocks elements. S-blocs are highly reactive. D-block are less reactive and more noble, especially the right-side metals such as Pt, Au. D-block metals form colored compounds. and s-block metals generally form colorless compounds.
Transition Metals vs Precious Metals
Precious metals are Au, Ag, Pt, Pd. These are a subset of transition metals with high economic value. They have extreme resistance to oxidation and corrosion, and high nobility. Most precious metals are transition metals, while not all transition metals are precious. For instance, some transition metals like Fe or Ni are base metals that oxidize easily.
Transition Metals vs Post-Transition Metals
All transition metals are d-block elements, but not all d-block elements are transition metals. Transition metals have partially filled d-orbitals. And post-transition metals have filled d-orbitals. They are Al, Ga, Sn, Pb and are located to the right of the d-block. Post-transition metals are softer and have lower melting points.
Conclusion
Transition metals are located in the d-block of the periodic table. These are defined by having partially filled d-orbitals. This makes them show variable oxidation states, form colored compounds, and act as catalysts. Transition metallic elements are generally hard, strong, and have high-density metals and melting points. They have excellent electrical and heat conductivity as well. Common transition metal elements are iron, copper, gold, silver, and titanium.
FAQ
What are the most useful transition metals?
The most useful transition metals are iron, copper, titanium,nickel, and platinum due to their high conductivity, strength, and alloying capability.
What's the difference between a metal and a transition metal?
All transition metals are also metals. They are a specific set of metals located in the center of the periodic table. The d-block metals have variable oxidation states, forming colored compounds, and acting as catalysts. While standard like alkali metals are generally softer, more reactive, and form colorless compounds.
Is aluminum a transition metal?
No, aluminum is not a transition metal. It is classified as a post-transition metal. It differs from transition metals because it lacks a filled d-orbital and shows fewer oxidation states.
Is zinc a transition metal?
Zinc is not a transition metal. It is classified as a post-transition. Because it has a completely filled 3d10electron shell in both its atomic (4S23,d10) and ionic (Zn2+,3d10) states.
 Tel/WeChat:
Tel/WeChat:  Email:
Email: 
 Home
Home
 What Is Ductility of Materials? Important Guide for Manufacturing
What Is Ductility of Materials? Important Guide for Manufacturing